In many popular medical journals and websites, the most proposed mechanism of red LED light therapy is mainly based on the light-sensitive responses in mitochondria. But what are the exact cellular signaling pathways after LED red light therapy? In this blog, we will look into a deeper investigation to reveal the cellular downstream induced by photobiomodulation red light therapy. Let's go on reading and see what researchers said about red and infrared light therapy.
How does red LED light therapy work?
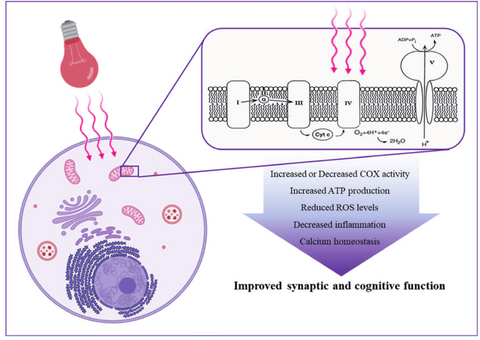
Red light therapy photobiomodulation (PBM), also known as low-level red light therapy (LLLT red light therapy), is the use of red and near-infrared light to induce healing processes, ease pain, and alleviate inflammation. The major chromophores have been determined as cytochrome c oxidase (Cox) in mitochondria and calcium ion channels (possibly intervened via light sensitive opsins). Secondary effects of photon absorption include enhanced ATP production, a temporal burst of reactive oxygen species, a release of nitric oxide, and calcium level modulation. Tertiary effects trigger a broad range of transcription factors, which leads to better cell survival, greater proliferation and migration, and novel protein synthesis. There is a notable biphasic dosage response as a feature of nir and red light therapy, with low amounts of light stimulating and high levels of light inhibiting. It has been verified that PBM light therapy prompts ROS production in healthy cells, but lowers ROS levels in oxidatively stressed cells in animal models of disease. PBM light therapy benefits also include the upregulation of anti-oxidant defenses to ameliorate oxidative stress.
The signaling pathways after LED red light therapy
TRP calcium channel proteins
Besides the most well-known notion of energy blueprint red light therapy through cytochrome c oxidase (Cox), it has lately become clear that another type of photoreceptor also plays an important role in signal transduction. These photoreceptors are thought to be members of the opsin family of light-sensitive G-protein coupled receptors (OPN). Opsins are proteins that are combined with light-sensitive chemicals for underlie vision, phototaxis, circadian rhythms, and other light-involved responses of organisms. Rhodopsin (OPN1) is the most well-studied opsin, and it is important for mediating vision in rod and cone photoreceptor cells in the mammalian retina. Other members of the opsin family (OPN2-5) are expressed in a variety of tissues throughout the body, including the brain, as documented in this study.

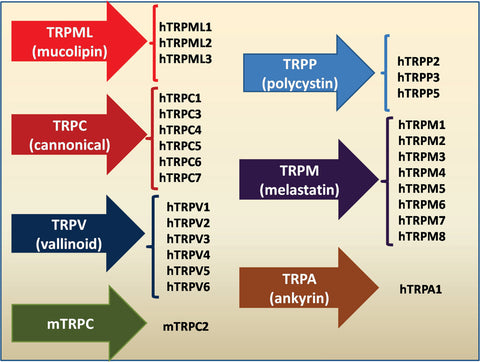
The opening of light-gated ion channels is one of the most well-defined signaling processes that happens following light-activation of opsins, which has been identified in this study. The members of the transient receptor potential (TRP) family of calcium channels, one of the best-defined signaling processes, are now recognized as pleiotropic cellular sensors that mediate responses to a wide range of external stimuli and are involved in a variety of cellular processes, which is studied in this research. TRP activation results in non-selective permeabilization to calcium, sodium, and magnesium (mostly of the plasma membrane). TRP channel proteins have been identified in most animals, organs, and cell types. There are seven related subtypes of the TRP channel family, including TRPC, TRPM, TRPV, TRPA, TRPP, TRPML, and TRPN.
Mast cells play a role in the biological effects of red laser light therapy on wound healing. Yang and colleagues observed that following cold laser red light therapy (532 nm), intracellular [Ca2+] increased, resulting in histamine release. The application of the TRPV4 inhibitor ruthenium red prevented histamine release, showing the importance of these channels in promoting histamine-dependent wound recovery following red light therapy for wound healing, which is the topic of this study.
TRPV1 ion channels appear to be involved in mast cell degranulation and laser-induced mast cell reaction. Infrared light (2,780 nm) inhibits capsaicin-induced TRVP1 activation in cultured neurons, reducing the production of pain sensations. TRPV4 is similarly inhibited by laser light therapy, but the antinociceptive impact is less than TRPV1 inhibition, as stated in this study.
Laser-evoked neuronal voltage fluctuation can be generated by neuron-stimulating effects from the pulsed infrared light therapy (1,875 nm). Nerve cells appear to be most actively responsive to infrared light irradiation, and TRPV4 channels were shown to be the principal effectors, as is verified in this study. Considering water is still the principal absorber in the infrared spectrum as the wavelength axis extends beyond 1500 nm these changes might be attributable to thermal effects in this study.
The increase in ATP
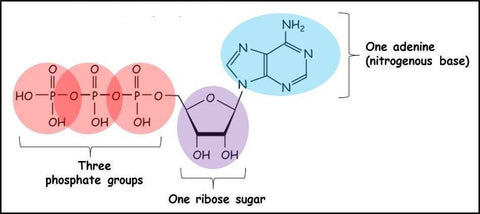
One of the most common and noteworthy observations following PBM, both in vitro and in vivo, is a rise in intracellular ATP. The enhanced activity of Cox contributes to the increase in ATP production. In this study, it was discovered that enhanced ATP production is observed following LED (850 ±20 nm and 630 ±10 nm) treatment in two muscles with different workouts (a predominately aerobic metabolism or a mixed aerobic and glycolytic metabolism). Extracellular ATP participates in a wide range of signaling pathways known as purinergic signaling. ATP purinergic signaling, working through P2Y G-protein-coupled receptors and P2X ligand-gated ion channels, is already identified as a non-adrenergic and non-cholinergic neurotransmitter, as in this study. Additionally, adenosine, a metabolic product of ATP, also performs a signaling function via the P1 G-protein-coupled receptor. Yet there is currently no evidence of red light therapy effectiveness for extracellular ATP or adenosine.
Bestqool: best at home red light therapy near me
With the exploratory nature of being a pioneer in the emerging medical phototherapy industry, Bestqool aims at providing our clients with the most effective and innovative solutions in medical-grade red light phototherapy. Years of engagement and research in this medical field allow Bestqool to deliver the best red light therapy at home devices for medical treatment to our clients, leading the industry with the highest safety standards and the design of superior optics.

- Bestqool, as an FDA-approved red light therapy device, guarantees the safety and efficacy of LED red light therapy for home use. All Bestqool devices are manufactured in our FDA-registered factories that adhere to ETL manufacturing requirements. We place a great value on the influence of our products on human health, as seen by the design of all Bestqool gadgets. All Bestqool red light treatment devices are EMF-free and equipped with a precise irradiance control, timing system, and cooling system to prevent heating damage.
- Bestqool invests much in research to provide the best LED light therapy at home equipment for optimal light therapy outcomes. Being proud of our innovative progress in LED manufacturing, Bestqool is able to deliver devices that focus on the most effective wavelengths in a small range with a high degree of irradiance control. Bestqool LED photon light therapy devices are backed by scientific evidence, an accomplishment of our professional knowledge in this field.
It can be said that the manufacturing technology of irradiance control is the most crucial in healing LED light therapy. Using 10 minutes of Bestqool light therapy devices is equal to 20 minutes of others. Bestqool also offers a three-year warranty for all Bestqool products, setting up the longest standard in this market for the best experience of natural red and infrared light therapy at home.











 Small
Small

 Moderate
Moderate

 Moderate
Moderate

 Moderate
Moderate

 Full
Full



